4Cell Nutri-T Medium 免疫细胞无血清培养基
Product Information:
4Cell® Nutri-T Medium: A Solution Without Serum
Cell-based immunotherapy is at the forefront of advanced cancer treatments. The most common cell-based immuno-therapies to date are T cell therapies (mainly CAR-Ts and TILs). Cells being used for immunotherapy are commonly cultured in media supplemented with human serum. The use of serum introduces further variability into the process due to donor-to-donor variation, which leads to inconsistent cell growth and characteristics. Eliminating serum simplifies the process, lowers the regulatory risk, and reduces the associated logistical burden. Nutri-T eliminates this need for serum addition by substituting serum‘’s critical components with specific proteins, lipids, and other small molecules.
Features
Xeno-Free, Serum-Free
Superior performance without serum addition
Exhibits excellent fold expansion and cell viabilty for TILs, CAR-T, PBMCs, etc.
Exceptional performance with cells starting at low initial seeding concentrations
Tested and developed on both healthy, and non-healthy patient derived donor cells
Formulation contains HSA and Phenol Red
4Cell® Nuti-T Cell Medium: Advancing Research and Clinical Applications
4Cell® Nutri-T is the ideal medium to use in the development and scale-up of cell-based therapeutic applications in the field of immune-oncology. Nutri-T is a xeno-free formulation demonstrating consistent and accurate results for both healthy donors (Fig. 1) and patient-derived (Fig. 2) T cells, without serum supplementation.
Figure 1: Nutri-T is Superior to Competitor Media in Expansion of Healthy PBMCs (With and Without CAR-T Transduction) at Multiple Seeding Densities
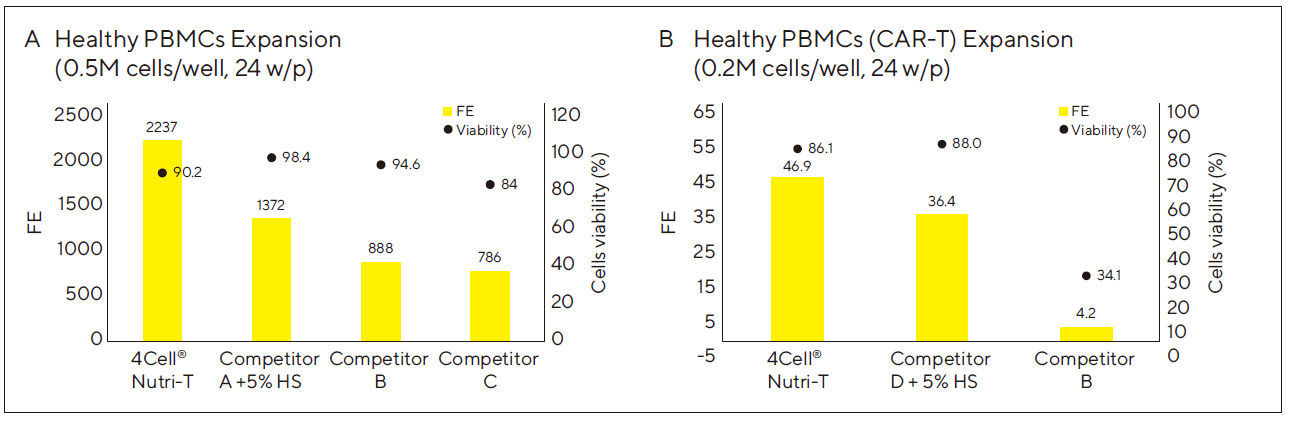
(A) 0.5M healthy donor PBMCs were seeded in 24w plates (2 ml media/well). Cells were activated with TransAct 1:100 and 600 IU/ml IL-2.
Cells were split and media renewed every 2 – 3 days. Fold expansion (FE) and cell viability were measured at Day 11.
(B) 0.2M PBMCs from healthy donors were seeded in 24w plates (2 ml media/well). Cells were activated with TransAct 1:100 and 600 IU/ml IL-2. 24 h. After seeding cells were transduced with a lentiviral vector expressing an EGFR-CAR-T. Cells were split and media renewed every 2 – 3 days. FE and cell viability were measured at Day 11.

4Cell® Nutri-T Medium: Excellent Performance With Patient-Derived Cancer T Cells
Most of the currently available xeno-free media for T cells have been validated only on cells isolated from healthy donor derived PBMCs, or healthy CAR-T manipulated cells. 4Cell® Nutri-T was developed in collaboration with the highly accredited Ella Lemelbaum Institute for Immuno-Oncology at Sheba Medical Center, Israel. The Sheba partnership allows Sartorius access to clinical, patient-derived TILs and T cells. This unique development platform resulted in 4Cell® Nutri-T medium exhibiting excellent performance even with clinical condition cells at low initial seeding concentrations (Fig. 2).
Figure 2: Nutri-T is Superior to Competitor Media in Expansion of Patient-Derived Cancer Cells for Both TILs and CAR-T Processes
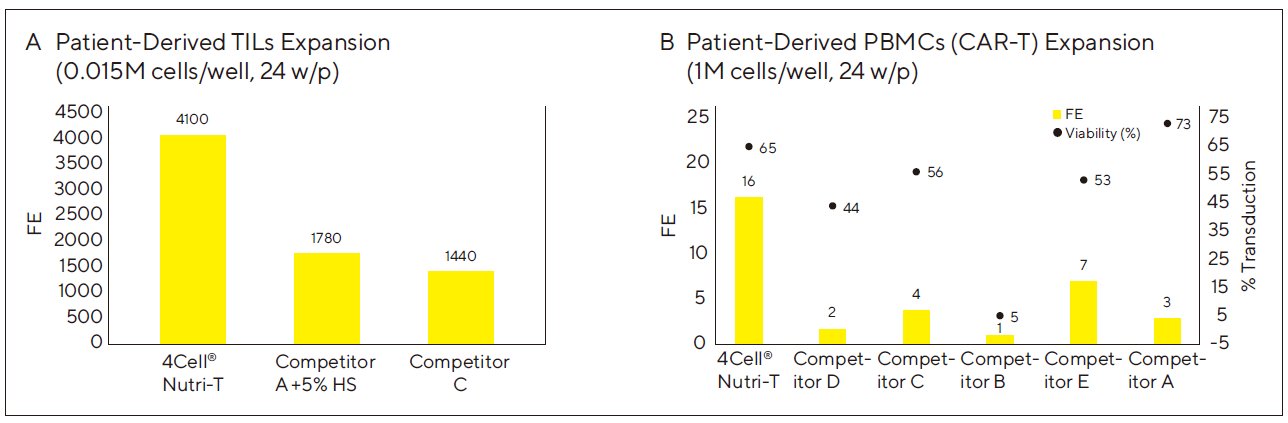
(A) TILs were isolated from a melanoma patient. 15,000 cells were seeded in a 24 well plate (2 ml/well) with PBMCs (1:100). Cells were activated with IL-2 (3,000 IU/ml) and OKT-3 (50 ng/ml). 2 ml and 4 ml of fresh medium + IL2 were added at days 5 and 7 respectively (total volume of 8 ml). Fold expansion was measured at 14 days. Inherent variations among primary T lymphocyte donor populations may result in varying outcomes.
(B) PBMCs were separated from peripheral blood of a lymphoma patient. Tested mediums were supplemented with 50 ng/ml OKT3 and 300 IU/ml IL2. At day 2 post seeding, 2 – 3M cells for the G-Rex24 were transduced with a CD19-CAR lentiviral vector in 6w/p pre-coated with RTN. Post transduction the cells were collected and reseeded. At day 4, 4 ml fresh medium +IL2 were added and at day 6, 50% medium was replaced with fresh medium + IL2. At day 9 transduction efficiency was evaluated and at day 10 Fold expansion was measured.
官网产品链接:4Cell® Nutri-T Medium国外产品链接
Copy right 2012 by Shanghai XP Biomed Ltd. All Rights Reserved
沪 ICP备12039761号-1 沪ICP备12039761号-2 沪ICP备12039761号-3 沪ICP备13002267号-1