VitroGel™3D 即用型水凝胶FAQ-Hydrogel Formation and Preparation Protocols
Hydrogel Formation and Preparation Protocols
Q How does gelation work in VitroGel?
A The hydrogel formation starts when VitroGel is mixed with the cell culture medium. The hydrogel molecules will interact with the ionic molecules, such as Ca2+ or Na+, from the cell culture medium to induce a matrix structure (hydrogel). For 3D culture operation, the hydrogel formation is in two stages: a) soft hydrogel formation, b) hydrogel stabilization.
Soft hydrogel formation: The hydrogel formation process is slow when a small amount of cell culture medium is used. At this stage, the hydrogel is soft and possess a shear-thinning and rapid recovering mechanical property, which makes the hydrogel injectable for in vivo The slow hydrogel-forming process and the injectable property of the soft hydrogel create a time frame for easy hydrogel transfer from the mixing tube to the cell culture plate.
Hydrogel stabilization: After soft hydrogel formation, adding additional cell culture medium on top of the hydrogel would allow more ionic molecules to penetrate the hydrogel matrix and further saturate the hydrogel cross-linking. A solid hydrogel would form during this process.
More information can be found here.
Q How do I adjust the hydrogel formation time?
A Adjusting the mixing ratio between the VitroGel solution and cell culture medium would change the hydrogel formation rate.
If the hydrogel solidifies too fast after mixing with the culture medium (showing as small solid gel chunk), adjust the mixing ratio by using less cell culture medium. For example, if mixing 2 mL hydrogel solution with 0.5 mL cell culture medium leads to solid gel chuck (particles), then mixing 2 mL diluted hydrogel solution with 0.2-0.4 mL cell culture medium would help to solve the issue.
On the other hand, if the hydrogel formation is too slow, adjust the mixing ratio by using more cell culture medium. For example, if mixing 2 mL diluted hydrogel solution with 0.5 mL cell culture medium leads to a slow hydrogel formation, then mixing 2 mL diluted hydrogel solution with 1-2 mL cell culture medium would help to solve the issue.
Q What can I do to prevent bubbles from forming when mixing the VitroGel solution with the cell culture medium?
A The bubble issue is related to the increased solution viscosity after mixing the gel solution with cell medium. Here are some suggestions that can help to reduce the formation of bubbles:
Warm up the VitroGel solution to 37° C to reduce the viscosity of the gel.
Gently mix the VitroGel solution with the cell medium. Then, pipette slowly without introducing bubbles.
Quickly spin the mixing tube to get rid of bubbles.
Q Can I add extracellular matrix proteins or other molecular compounds into VitroGel?
A Yes. Extracellular matrix proteins or other molecular compounds can be added into the VitroGel system. Before hydrogel formation, add the proteins or the molecular compounds into the cell culture medium and then mix directly with the VitroGel 3D hydrogel solution. Please note that the hydrogel formation time and the final gel stiffness might change due to the salts contained in the proteins or chemical compounds. Please contact us at support@thewellbio.com if you have any questions or concerns about adding additional compounds to the VitroGel system.
Q Why does the hydrogel sticks loosely to the tissue culture plate?
A This issue might be because of the following reasons:
Using a non-treated tissue culture plate, which has a more hydrophobic surface. This reduces the attachment of hydrogel/cells on the surface of the well-plate. For better performance, we suggest using a treated tissue culture plate with VitroGel.
Adding the hydrogel as a dome instead of covering the whole bottom of the well plate might also cause this issue. We suggest gently tilting and swirling the well plate after adding the hydrogel to ensure the whole bottom of the well plate is covered by the gel.
Not waiting long enough before adding the additional medium on the top of the hydrogel. After transferring the hydrogel to the well plate, please wait 10-30 minutes for hydrogel stabilization before adding the top medium. Adding the medium before the hydrogel stabilizes would disrupt the structure of hydrogel. The lower the concentration of the hydrogel, the longer the waiting time is needed.
After the initial soft hydrogel formation, it is crucial to make sure the hydrogel is stable and attached to the bottom of the well plate before adding the cover medium. If the hydrogel is not stable, it might detach from the bottom of the well after adding the cover medium. During the hydrogel formation, the gel is soft; do not shake the plate or position the plate vertically. Keep the plate horizontally.
Q Can I form a dome shape culture?
A Adding the hydrogel as a dome shape is not recommended. The dome shape may not stick to the bottom of the culture plate for long term culture. The hydrogel should cover the whole bottom of the well plate. We suggest gently tilting/swirling the well plate after adding the hydrogel to ensure the gel coats the entire bottom of the well plate. Using a 96 well plate can reduce the usage volume of the hydrogel to 30-75 µL/well. There is no issue with molecular penetration when adding the cover medium to feed the cells.
Q Can I use a serum-free medium with VitroGel?
A Yes, serum-free medium works with VitroGel.
Q Is it possible to dilute VitroGel hydrogels with cell culture medium instead of the dilution solution?
A For the High Concentration hydrogels, it is not recommended. The concentration of the ionic molecule in medium would be much higher than the dilution solution, so diluting VitroGel with cell culture medium may cause the gel to form too quickly and chunky. We recommend using VitroGel Dilution Solution to adjust the highly concentrated VitroGel.
Alternatively, VitroGel Hydrogel Matrix (Cat# VHM01) is a better option for scientists who do not want to worry about dilution and only want to mix the hydrogel with cell suspension directly.
Q How fast should I transfer the sample from the mixing tube to the culture plate?
A After mixing with the cell culture medium, we recommend transferring the mixture to the tissue culture plate within 10 minutes. The hydrogel formation starts after mixing the VitroGel solution with the cell culture medium.
If you have multiple samples with different hydrogel conditions or cell types to prepare, we recommend transferring the mixture of sample 1 to the tissue culture plate before mixing the hydrogel with cell culture medium for sample 2.
Q Can I place the gel mixture in an incubator instead of leaving it at room temperature for gel stabilization?
A Yes, it is fine to place the gel mixture in an incubator instead of room temperature, but please note, the gel forms a little slower in an incubator than at room temperature. As a result, for lower concentrated gels, you will need to keep it in an incubator for a little longer. However, for gels at a higher concentration or VitroGel Hydrogel Matrix, it is not a problem.
Q Can I pre-coat the culture plate to increase hydrogel attachment?
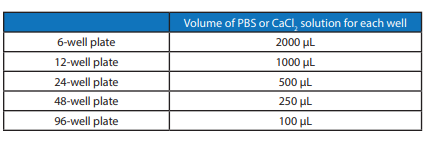
A Although, for most cases, pre-coating the culture plate is not necessary, coating the plate with 1X PBS or 10-100 mM CaCl₂ can improve the hydrogel attachment. Please check the following protocols for coating the culture plate:
Add the PBS or CaCl₂ solution to the culture plate for 30 min.
Remove the PBS or CaCl₂ solution, open the lid under the biosafety hood for 10-20 min before adding the hydrogel. The recommend volumes of PBS or CaCl₂ for different sizes of well plates are list in Table 1 below.
Table 1: Recommend volume of PBS or CaCl₂ solution for pre-coating well plate
Copy right 2012 by Shanghai XP Biomed Ltd. All Rights Reserved
沪 ICP备12039761号-1 沪ICP备12039761号-2 沪ICP备12039761号-3 沪ICP备13002267号-1